| ESA | Return to https://www.halos.com/book/ctm-app-05-b.htm. | ESA |
< Prev T of C ... 12 13 14 15 Epi. Cat. App. Ref. Cred. Next >
The coloration extinction boundary is poorly defined near threshold coloration; only a few very light bands in biotite could be reliably measured. Reproducible measurements were obtained in the plateau region (14), where variations in band size are minimal. Darker halos in biotite generally have slightly larger radii than lighter halos (3, 4). Also, reversal effects in some biotites immediately exterior to the terminus of a halo ring cause apparent diminution of the radius. Therefore, while there are differences between the sizes of medium coloration hands (Table 1, column 2) and the radii of U halos in biotite (Table 1, columns 8, 9, and 10) that could be interpreted in terms of an actual change in Eα and λ (16), such differences more likely arise from a combination of dose and reversal effects (15, 17), producing slightly diminished radii. Diminution of U halo radii may also result from attenuation of α-particles within the small but relatively dense zircon radiocenters. Even though slight differences between band sizes and U halo radii do exist in biotite, the idealized U halo ring structure (Fig. 1a) compares very well with an actual U halo in biotite (Fig. 1f).
Biotite and fluorite are good halo detectors, but fluorite is superior because the halo rings exhibit more detail, often have smaller radiocenter diameters (< 1 μm), and have almost negligible size variations due to dose effects in the embryonic to normal stages of development. Figure 1g shows an embryonic U halo in fluorite with only the first two rings fully developed; the other rings are barely visible because, due to the inverse square effect, threshold coloration has not been reached. Figure 1h shows a U halo in fluorite in the normal stage of development, when nearly all the rings are visible. This halo closely approximates the idealized U halo in Fig. 1a. Under high magnification even separation of the 210Po and 222Rn rings may be seen. Figure 1i shows another U halo in fluorite, with a ring structure that is clearly visible but not adequate for accurate radius measurements.
In Table 1, columns 4, 11, and 12, the fluorite band sizes agree very well with the U halo radii measured in this mineral by myself and Schilling (9). This suggests that the differences between U halo radii and band sizes in biotite are not due to a change in Eα However, experimental uncertainties in measuring U halo radii preclude establishing the constancy of λ to within 35 percent, and under certain assumptions U halos provide no information at all in this respect (16).
While halos with point-like nuclei which show well-defined, normally developed rings (as in Fig. 1h) can be used to determine the Eα's of the radionuclides in the inclusion, there are pitfalls in ascertaining what constitutes a normally developed ring. In contrast to the easily recognizable U halos in fluorite in Fig. 1, g to i, the overexposed fluorite U halo in Fig. 1j shows a diminutive ghost inner ring, which could be mistaken for an actual 238U ring. Figure 1k shows two other partially reversed U halos, one of which shows the diminutive inner ring, while in the other all the inner rings are obliterated. The U halo in Fig. 1l is even more overexposed, and encroaching reversal effects have given rise to another ghost ring just inside the periphery. Figure 1m shows a still more overexposed U halo; in which second-stage reversal effects have produced spurious ghost rings that are unrelated to the terminal α-particle ranges.
Since this association of the halos in Fig. 1, l and m, with U α-decay cannot be easily proved by ring structure analysis alone, I have utilized electron-induced x-ray fluorescence to confirm this identification. Figure 3a shows the prominent Ca x-ray lines of the fluorite matrix (the F lines are below detection threshold) along with some background Ag and Rh lines which are not from the sample, but are produced when back-scattered electrons strike a Ag-Rh alloy pole piece in the sample chamber. Figure 3b, the x-ray spectrum of a halo radiocenter typical of the halos in Fig. 1, l and m, clearly shows the x-ray lines due to U (as well as a small amount of Si) in addition to the matrix and background peaks. A more detailed analysis (18) reveals that the Uζ line masks a small amount of Pb probably generated by in situ U decay.
The variety of U halos shown in Fig. 1, g to m, establishes two points: (i) only a thorough search will reveal the numerous variations in appearance of U halos, and (ii) unless such a search is made, the existence of halos originating with α-emitters other than 238U or 232Th could easily be overlooked.
So far, three criteria have been used to establish the identity of U halos: (i) close resemblance of actual halos in biotile (Fig. 1f) and fluorite (Fig. 1h) to the idealized ring structure [p. 242] (Fig. 1a), (ii) identification of lines in x-ray fluorescence spectra, and (iii) agreement between U halo radii and equivalent band sizes (very good in fluorite and fair in biotite and cordierite). Using the third criterion (either band sizes or U halo radii) I can determine Eαfor a normally developed fluorite halo ring to within ± 0.1 Mev. For biotite halos, U halo radii may form a suitable standard for determining Eα for rings that show reversal or other effects characteristic of U halos in the same sample. If good U halos are not available, and if the halos with variant sizes show well-developed rings without reversal effects, then the band sizes form a suitable standard for Eα determination when coloration intensities of variant halos and band sizes are matched.
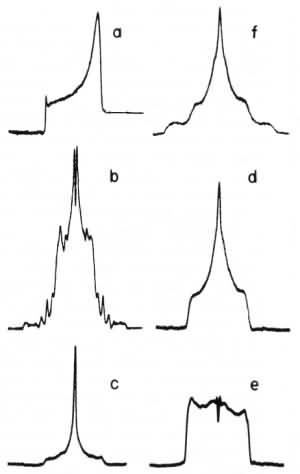 Fig. 2. Densitometer profiles of the photographic negatives of (a) Fig. 1e, (b) Fig. 1f, (c) the light 210Po halo in Fig. 1n, (d) the medinm 210Po halo in Fig. 1n, (e) the dark 210Po halo in Fig. 1n, and (f) Fig. 1p. |

|
Doc.: T of C ... #1 #2 #3 #4 #5 #6 #7 #8 #9 ...
< Prev T of C ... 12 13 14 15 Epi. Cat. App. Ref. Cred. Next >
© 2004
Earth Science Associates