| ESA | Return to https://www.halos.com/book/ctm-app-06-b.htm. | ESA |
< Prev T of C ... 12 13 14 15 Epi. Cat. App. Ref. Cred. Next >
The ion-microprobe mass spectrum of the mica matrix surrounding the 'spectacle' halo was nearly identical to the mica spectrum shown in Fig. 2 and is not repeated in Fig. 3. Figure 3 (top centre) shows the portion m/e = 160−264 of the ion-microprobe spectrum (verical log scale) of several of the inclusions. Also shown is the actual ion-probe trace of the important region from m/e = 204−210 using a linear vertical scale and an expanded horizontal scale. There is no significant ion current above m/e = 209; that is, no significant ion signals were detected at any of the prominent U and Th peaks: 238(U+), 254(UO+), 232(Th+) and 248(ThO+). No m/e = 204 was detected above background (1 c.p.s.), and the 206/207 mass ratio was 20 (206 signal 2,000 c.p.s.).
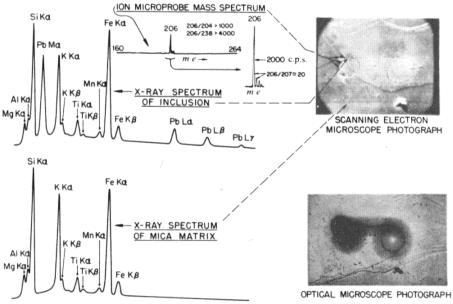 |
| Fig. 3 Ion microprobe and SEMXRF spectra of mica matrix and 210Po halo inclusions. |
Figure 3 also shows SEMXRF spectra of the surrounding mica and of one of the Po halo radiocentres. Lead is the only element detectable in this radiocentre exclusive of the mica; some adjacent radiocentres revealed Bi as well. The use of two different instruments, and longer counting times, account for the slightly different X-ray spectra in Figs 2 and 3. The excellent resolution of the SEM showed the Pb-rich areas to coincide exactly with the Po halo radiocentres which are visible both in ordinary transmitted (Fig 1) and reflected light microscopy. Regions as close as 1 μm to the radiocentres showed virtually no Pb or Bi, implying little if any diffusion loss from the inclusions.
As the X-ray data definitely show Pb (and sometimes Bi) in the 'spectacle' halo radiocentres, and as there is no evidence for any molecular ion contribution in the region from m/e = 204−238, [p. 245] the 206, 207 and 208 peaks are interpreted as Pb isotopes and 209 as 209Bi. 204Pb, a constituent of both common and primordial Pb, is missing (no 204 peak), implying that the 'spectacle' halo inclusions analyzed contained no detectable Pb of either of these types. Absence of the 232, 235 and 238 peaks is interpreted as showing the inclusions contain virtually no 232Th, 235U or 238U and, therefore, no radiogenic 208Pb, 207Pb or 206Pb derived from the in situ decay of these isotopes. The 207 and 208 [p. 246] peaks are therefore attributed to 207Pb and 208Pb, perhaps arising from the decay of minute amounts of 211Bi and 212Bi within the inclusions5,6. The 209Bi is considered to be primordial.
The outstanding feature of the mass analysis is the prominent 206 signal which, when attributed to the presence of 206Pb in the inclusions, fits perfectly with the prediction based on ring structure measurements, that is, that the 206Pb is radiogenically derived, not from U or Th, but directly from 210Po α decay. In this respect, the large difference in the 206/238 (206Pb/236U) ratio between the 'spectacle' halo and the U−Th halo (Figs 2 and 3) is especially significant. Clearly the 'spectacle' halo resulted from 210Po α decay; an explanation for its geometry is still under study.
Because the Pb isotope in these inclusions is not explicable as any combination of common, primordial, or from in situ Pb derived radiogenically in situ from U or Th, we conclude that a different type of Pb, derived from Po α decay, exists in nature. Supportive evidence comes from electron-probe and ion-probe analyses of a 218Po halo radiocentre found in a mica from the Iveland District, Norway, which yielded a 206Pb/207Pb ratio of 23. This is consistent with that expected from 218Po a decay to 206Pb. Such a Pb ratio is impossibly high based on normal isotopic 238U/235U decay, the theoretical maximum being 21.8.
Other investigations have shown varying mixtures of U-derived and Po-derived Pb may occur in the same radiocentre, for there exists an almost continuous halo spectrum stretching from "pure" U to "pure" Po haloes. Only a few (<0.01) Po haloes in biotite may survive the delicate sectioning process necessary for SEM X-ray analysis.
Just as important as the existence of a new type of lead is the question of whether Po haloes which occur in a granitic or pegmatitic environment (for example, in mica, fluorite or cordierite) can be explained by accepted models of Earth history1. (R. V. G. has found other 210Po haloes that differ essentially from those in granites-unpublished information.)
This research has been sponsored by the United States Atomic Energy Commission under contract with Union Carbide Corp. and by Columbia Union College with an assistance grant from the National Science Foundation. Thanks are due to R. I. Gait and J.A. Mandarino, Royal Ontario Museum, Louis Moyd, National Museum of Canada, and G. Switzer, United States National Museum, for providing specimens.
| ROBERT V. GENTRY |
| Chemistry Division, Oak Ridge National Laboratory, |
| L. D. HULETT |
| Analytical Chemistry Division, Oak Ridge National Laboratory |
| S. S. CRISTY J. F. MCLAUGHLIN |
| Laboratory Development Department, Oak Ridge Y-12 Plant, Oak Ridge, Tennessee 37830 |
| J. A. MCHUGH |
| Knolls Atomic Power Laboratory, Schenectady, New York 12301 |
| MICHAEL BAYARD |
| McCrone Associates, 2820 Michigan Street, Chicago, Illinois 60616 |
Received July 31, 1974.
References
- Gentry, R. V., Science, 184, 62 (1974).
- Kohnan, T. P., Ann. N. Y. Acad Sci., 62, 503 (1956).
- Henderson, G. H., Mushkat, C. M., and Crawford, D. P., Proc. R. Soc., A158, 199 (1934); Henderson, G. H., and Turnbull, L. G., ibid., 145, 582 (1934); Henderson, G. H., and Bateson, S., ibid., 573 (1934).
- Gentry, R. V., Ann. Rev. NucI. Sci., 23, 347 (1973).
- Gentry, R. V., Cristy, S. S., McLaughlin, J. F., and McHugh, J. A., Nature, 244, 282 (1973).
- Gentry, R. V., Science, 173, 727 (1971).

|
Doc.: T of C ... #2 #3 #4 #5 #6 #7 #8 #9 #10 ...
< Prev T of C ... 12 13 14 15 Epi. Cat. App. Ref. Cred. Next >
© 2004
Earth Science Associates