| ESA | Return to https://www.halos.com/book/ctm-app-07-b.htm. | ESA |
< Prev T of C ... 12 13 14 15 Epi. Cat. App. Ref. Cred. Next >
However, in this matrix the situation is quite different. A solution-permeated wood in a gel-like condition would exhibit a much higher transport rate as well as unusual geochemical conditions which might favor the accumulation of 210Po and 210Pb nuclides. Evidence that this accumulation was essentially finished prior to complete coalification comes from the fact that most Po halos are plastically deformed; furthermore, after coalification it is much more difficult to account for such rapid and widespread migration of the radionuclides (that is, within the 210Po half-life). For example, a hundred or more 210Po halos are sometimes evident in a single thin section (2 cm by 2 cm) of coalified wood, and they occurred quite generally in the thin sections examined (11). Of the thousands of Po halos seen in this matrix, only three show any trace of a ring that could possibly be attributed to 214Po α-decay [that is, from the accumulation of the U-daughters 214Pb (t1/2 = 27 minutes), 214Bi (t1/2 = 20 minutes), or 214Po (t1/2 = 164 μsec)], and none has been seen with a ring from 218Po α-decay [that is, from the accumulation of short-lived 218Po (t1/2 = 3 minutes)]. (Possibly these faint outer rings are of chemical rather than radioactive origin.)
Positive identification for the 210Po halos comes from the IMMA analyses. Compared to a 238U halo radiocenter. a 210Po halo inclusion should contain much less 238U (perhaps none at all) and much more of the 210Po decay product 206Pb. The IMMA analyses of Po halo inclusions showed that the 238U content was low, the 238U/206Pb ratios varying from 0.001 to 2.0. [These values were corrected for the different ionization efficiencies (~ 2 : 1) of Pb+ and U+ in this matrix.] This small 238U content implies that only an extremely small amount of Pb could have been generated by in situ U decay. There are certainly three other possible sources for the Pb in these inclusions: (i) common Pb, (ii) Po-derived radiogenic Pb generated by in situ decay of secondarily accumulated 210Pb and 210Po, or (iii) U-derived "old" radiogenic Pb that had accumulated in the hypothesized (12) Precambrian U ore deposit (which is one possible source of the U now in the Colorado Plateau) prior to the time it was carried with the U in solution into the wood. Since the 204Pb count rates, which are unique indicators of common Pb, ranged from undetectable to a few counts per second above background when 206Pb count rates were several thousand counts per second, it was evident that relatively little common Pb was present. Thus only 206Pb/207Pb ratios had to be measured to obtain evidence of 206Pb originating from the decay of 210Po: the results were indeed confirmatory.
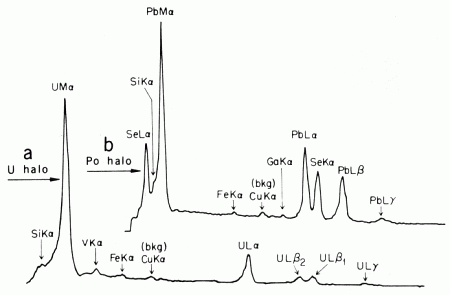 Fig. 2. Curve a, EMXRF spectrum of a U-rich radiocenter. Curve b, EMXRF spectrum of the radiocenter of a 210Po halo. |
The ratios obtained were as follows: 206Pb/207Pb = 8 ± 0.5, 11.6 ± 0.3, 11.7 ± 0.4, 13.3 ± 0.7, 13.4 ± 1.0, 13.7 ± 0.6, 13.9 ± 0.6, 14.8 ± 0.9, 15.8 ± 1.1, and 16.4 ± 0.5. The variation in this ratio can easily be understood to have resulted from the addition of an increment of 206Pb (generated by in situ 210Po decay) to the isotopic composition of the "old" radiogenic Pb. The lowest Pb ratio, obtained from a very lightly colored 210Po halo, differs slightly from the lowest Pb isotope ratio previously determined on bulk samples of Colorado Plateau U ore specimens (12).
What is the meaning of these Po halos? Clearly, the variations in shape can be attributed to plastic deformation which occurred prior to coalification. Since the model for 210Po formation thus envisions that both 210Po and 210Pb were accumulating simultaneously in the Pb-Se inclusion, a spherical 210Po halo could develop in 0.5 to 1 year from the 210Po atoms initially present and a second similar 210Po halo could develop in 25 to 50 years as the 210Pb atoms more slowly α-decayed to produce another crop of 210Po atoms. If there was no deformation of the matrix between these periods, the two 210Po halos would simply coincide. If, however, the matrix was deformed between the two periods of halo formation then the first halo would have been compressed into an ellipsoid and the second halo would be a normal sphere. The result would be a dual "halo" (Fig. 3c). The widespread occurrence of these dual halos in both Triassic and Jurassic specimens (13) can actually be considered corroborative evidence for a one-time introduction of U into these formations (1, 2), because it is then possible to account for their structure on the basis of a single specifically timed tectonic event. The fact that dual halos occur in only about 1 out of 100 single Po halos is of special significance (14).
In halos with U radiocenters, the low Pb abundance made it generally quite difficult to measure U/Pb ratios with EMXRF (Fig. 2a) techniques. More sensitive IMMA measurements on these U radiocenters revealed 238U/206Pb ratios (15) of approximately 2230; 2520; 8l50; 8300; 8750; 18,700; 19,500; 21,000; 21,900; and 27,300 (again corrected for different ionization efficiencies). Typically, the U+ ion signals from which these ratios were derived were greater than 3 × 104 counts per seconds (cps); for example, the 19,500 value was obtained from a halo with a U+ signal of 106 cps (± 5 percent) with background 3 cps. We checked the 238U/235U ratio independently (and found it normal) by excising several radiocenters and analyzing them directly on the filament of a high sensitivity thermal ionization mass spectrometer (16).

|
Doc.: T of C ... #3 #4 #5 #6 #7 #8 #9 #10 #11 ...
< Prev T of C ... 12 13 14 15 Epi. Cat. App. Ref. Cred. Next >
© 2004
Earth Science Associates